RLYB116
RLYB116 Program
- Stage
- Phase 1
- Indication
- Complement-mediated diseases, including PNH, gMG, and APS
- Approach
- Novel antibody mimetic fusion protein designed to inhibit Complement Factor 5 (C5)
- Mode of Administration
- Subcutaneous injection
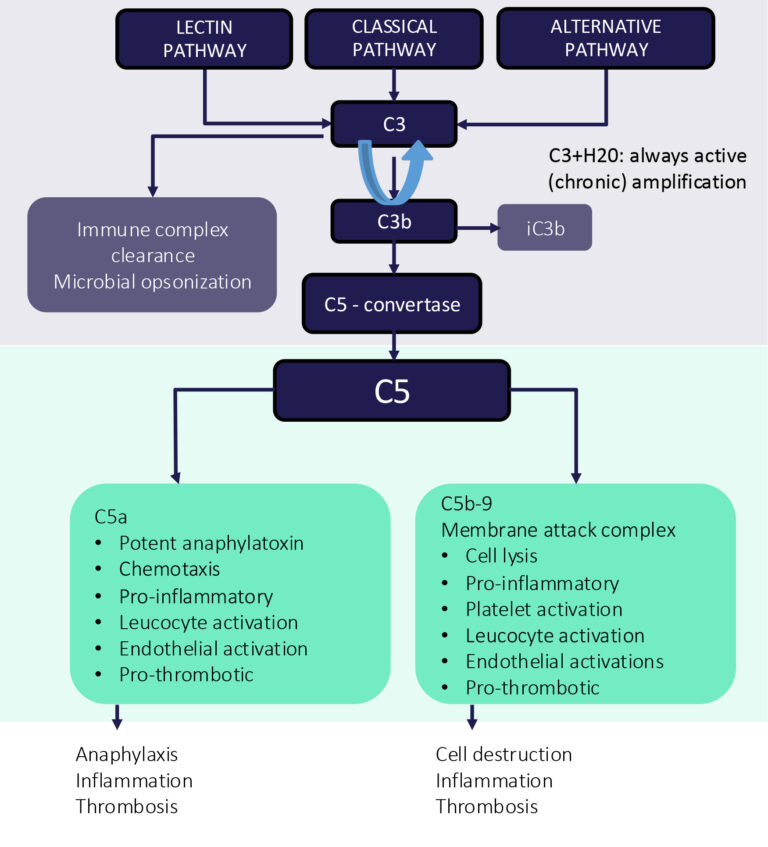
Complement Dysregulation
The complement system plays a central role in innate immunity, as well as in shaping adaptive immune responses. Dysregulation of the complement pathway has been linked to a broad range of diseases, including paroxysmal nocturnal hemoglobinuria (PNH), antiphospholipid syndrome (APS), and generalized myasthenia gravis (gMG), reinforcing its importance as a target for therapeutic intervention.
RLYB116 is a novel, innovative, once-weekly, small volume, subcutaneously injected C5 inhibitor designed to meet patient demand for a convenient, self-administered at-home solution. RLYB116 is an antibody mimetic fusion protein composed of two linked components:
- An Affibody® molecule with high affinity for C5, thereby inhibiting terminal complement activation, and
- Albumod™ technology that enables binding to serum albumin for the potential of an extended plasma half-life and broad tissue distribution
C5: A Proven Target in Complement Dysregulation

“RLYB116 presents a unique opportunity for the Rallybio team to utilize its deep experience in successfully designing, developing, and delivering complement inhibitors for patients with rare diseases.”
—Steve Ryder, M.D., Chief Medical Officer